
Picture # 1

Picture # 2
Picture # 3
Picture # 4

Picture # 5

Picture # 6

Picture # 7

Picture # 8

Picture # 9

Picture # 10

Picture # 11
by Kelly Ross, A.G. (C.I.G.), Lethbridge, Alberta
The Canadian Diamond industry is in its infancy, however, the size and relative value of this industry may soon be among the largest in the world. Aside from scattered small deposits, there are no other known valuable gem deposits in Canada. The mine to market chain for diamonds has many steps including the original mining of the rough gem material. The sale of the rough gem material to large buyers. The cutting of the rough diamond into consumer material. The sale of the cut diamonds to wholesalers and then the sale of diamonds from the wholesalers to retailers. Lastly the sale of diamonds from retailers to consumer. Until recently, the diamond industry in Canada virtually existed in the last two steps of the chain. With the mining of diamonds in Canada that is changing and all the steps of mine to market chain are evolving.
In order to deal effectively with diamonds, one should be well educated in the identification and evaluation of the cut and polished diamonds. A good working knowledge of diamonds in their natural form, known as ěroughsî is very desirable. With the growth of the diamond industry in Canada this knowledge will be increasingly more important to Canadians in the gem trade.
The following document discusses in general gem quality rough diamonds covering in brief, rough diamonds in terms of;
- Recovery of Rough Diamonds
- Structure and General Shapes
- Surface features
- Inclusions and Color
- Method of Identification.
Recovery of Rough Diamonds
Diamond recovery is important in understanding and identifying diamonds as the process of recovering diamonds can help identify a country of origin. The two recovery processes are that of Pipe or Mine deposits and Placer deposits.
Pipe or Mine deposits are found in the source rock itself and the rock that the diamonds formed in is crushed to free the diamonds within it. At present this is how diamonds are mined in Canada. The source rock is known as kimberlite and in simple terms is a volcano that bust to the surface of the earth cutting through several layers of rock to reach the surface. In doing so it carried chunks of rock from the layers that it cut through and within these chunks of rock are found the diamonds. The kimberlite cools and what is left is a ěpipeî that extends in the shape of a cone into the earth, with the wide end of the cone at the surface. The diamonds in these mines have not been exposed to erosion or weathering process and as a result are typically angular in shape showing classic crystal shapes with clearly defined faces and edges. However, in limited cases diamonds may have been exposed to radiation and this can alter the appearance of the diamond in terms of its shape and surface features. As a further note there is another type of commercially viable source rock known as Lamphorite that like kimberlite can contain diamonds. Diamoniferous lamphorites of commercial viability are much more rarer than kimberlite however some exist including that at the Australian Argyl Mine.
Placer deposits are deposits of diamonds which have been moved from their original source rock to another place through the process of erosion and other forces of nature. Often Placer deposits are found in river beds, on beaches, or sea floors. The diamonds in these cases have been freed from the Kimberlite through erosion and transported by wind, water or gravity to a likely resting place in a river bed, sea floor or beach. Millions of years of erosion of the kimberlite remove enough diamonds that a concentration of them can build up in another place, forming a placer deposit. Although diamonds are the hardest mineral it is subject to weathering as is any other mineral. As the diamond is transported from the kimberlite pipe it may be scratched or bumped along the way. Depending on the terrain, the distance the diamond is moved, and the time involved, the diamond may have its edges worn down and rounded like any other stream pebble. A diamond found in a placer deposit will show varying degrees of rounding from very slight to that similar to polished round stream pebble.
The potential for placer deposits of diamonds in Canada are unlikely as recent glaciation would not lend itself to the formation of placer deposits due to the limited time that has gone by since the last glaciation. Placer deposits formed prior to the glacial period would likely have been scraped away by the glaciers and scattered over such a large area that although a single gem may be found it is extremely unlikely that a second one would be found in the vicinity. The exception perhaps would be within glacial till deposits which are concentrations of glacial sediment that could be a source of a placer deposit. The writer is unaware of any placer deposits of gem quality diamonds in Canada.
The following is a list of diamond producing countries and generalized recovery types.
South Africa Pipe and Placer
Zaire Pipe and Placer
Namibia Placer (beach)
Angola Placer (river)
Ghana Placer (river)
Guinea Placer (river)
Ivory Coast Placer (river)
Liberia Placer (river)
Sierre Leone Placer (river)
Russia Pipe and Placer (minor deposit)
Australia Pipe
Canada Pipe
Given the forgoing, in general it could be said that gem quality diamonds produced in Canada originate in Pipe Mines and therefore the roughs would appear angular and have a marked absence of weathering.
Structure and General Shapes of Rough Diamonds
Structure
Diamond is quite unique as it possesses qualities that make it easily stand out from other minerals. It is widely known that diamond is the hardest natural substance known having a Hardness Value of 10 on the Mohs hardness scale of 1-10. One(1) being the softest and ten(10) being the hardest. This hardness reflects the ability to penetrate or scratch the surface of the gem. Despite being the hardest mineral, Diamond is rather brittle. That is, it can be ěcrackedî rather easily if it is struck in a direction of its cleavage planes and pieces can be broken off. The difference between hardness and brittleness was confused in early diamond mining in South Africa and many a diamond was crushed or shattered by a miner who use to hit the gems with a sledge hammer to test for a gems authenticity. The belief was if the gem is the hardest mineral it could withstand the blow of a sledge hammer and thus be a real diamond, whereas other substances would be crushed like ordinary rock. Consequently, many authentic diamonds were mistaken for ordinary rock when they split or ěcleavedî upon being hit with sledge hammer or reduced to many smaller pieces.
Another property of diamond is that it is highly thermal conductive. It is extremely effective at conducting heat as a result of the carbon material its formed from and its highly organized atomic structure.
Diamond also has a distinctive feature in its weight, known as Specific Gravity. Specific Gravity is the weight of a material divided by the weight of an equal volume of water it displaces. ěDiamond has a specific gravity of 3.5 which is about a third more dense than typical rock.î Diamonds and the Northwest Territories, Canada pg2. The Specific Gravity of diamond is precisely measured to 3.52 .
Diamond falls into the cubic crystal system also known as Isometric. The mineral itself is a hard dense crystalline form of carbon. Carbon can exist in many forms aside from diamond including the more familiar, graphite, which is found in the common pencil. When carbon is compressed under high pressure and elevated temperatures the conditions exist for it to turn into the form of carbon known as diamond. These conditions needed to produce diamond are found within the earths crust at depths of up to 150km below the surface. Man has also been able to mimic these conditions and has produced man-made diamonds. These man-made or synthetic diamonds are the same in composition however display subtle differences in structure and properties that distinguish them from their natural counterparts. As well when they are created they look quite different than rough diamonds. Its when they are cut and polished do they appear like natural diamonds that are cut and polished.
Each atom of carbon in a diamond has the same shape. The individual atoms of diamond are arranged in an orderly pattern and their shape is reflected in the external shape of the diamond.
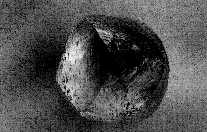
This external shape may be displayed in several ways however, the predominant shape seen is referred to as the gems ěhabitî. The predominant habit (shape) of the rough diamond is that of an octahedron. The classic octahedron appears like 2, four sided pyramids stuck together at their base, with the sides of the pyramid on top basically lining up with the sides on the bottom.
Shapes
Octahedron
As mentioned above, the predominant shape of rough diamonds is that of the octahedron. (Image#1).
Dodecahedron
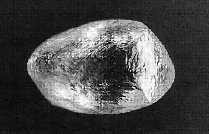
This is also a very common shape of rough diamond. It has 12 faces that give the rough diamond of this shape a less angular appearance than the octahedron. The faces on the dodecahedron as smaller and there are more of them and can give the appearance that the gem ěbulging outî. ěThese (octahedrons and dodecahedrons) shapes are most commonly encountered in rough gem diamonds and represent approximately 50% of rough diamonds in a typical parcel.î Rough Diamonds pg 22. (Image#2)
Cubes
Diamonds may actually occur in the shape of a cube. This seems natural as diamond is in the cubic crystal system. However, the occurrence of gem quality diamonds in a cube shape is rather rare. ěIt does occur in gem quality form occasionally but much more frequently in industrial (non-gem quality) or non cuttable form.î Rough Diamonds pg 14. The cube surface may be concave or convex as may be the face junctions (ribs).
Whole and Irregular Shapes
Rough diamonds that are modified forms of the octahedron, dodecahedron and cube and display both rounded faces, points and ribs are referred to as ěwhole diamondsî. If the same conditions prevail and the rough is elongated its known as an ěirregularî. These shapes are common to river bed and alluvial placer deposits reflecting the rounding of edges that occur through weathering and transport from their surface origin to the place where their found as a placer deposit.
Whole diamond (Image #3) from Rough Diamonds pg16, Irregular diamond (Image #4) from Rough Diamonds pg 16.
Cleavages
These are rough diamonds of any shape that have a broken surface that may or may not be in a cleavage direction. The cleavage plane in a diamond is parallel to the faces of the octahedron, regardless of its exterior appearance. Although diamond is the hardest known natural substance it can be relatively easily broken along its cleavage plane. This characteristic is exploited by persons that cut and polish diamonds to easily remove unwanted parts off the rough diamond. Rather than having to saw through the rough diamond, they can ěcleaveî the diamond with tools that basically amount to a hammer and chisel.
When a cleavage plane is recognized, it is a identifying feature of a rough diamond and also lends insight into what the rough looked like prior to having lost a piece along the cleavage plane.
I have used the mineral fluorite to illustrate a cleavage plane. Fluorite is also a mineral in the cubic crystal system that forms as an octahedral with cleavage planes the same as diamond. I used fluorite as it significantly cheaper than diamond and cleaves quite easily. Image #5 shows the octahedral shaped fluorite crystal prior to being cleaved. Image #6 shows the fluorite crystal after being cleaved and the fragment that broke away. Notice the cleavage surface parallel to the octahedron face.
Flats
The term describes a rough that is shallow in depth with little or no body and the shape often does not lend itself to easily being cut into a retail diamond. Typical flats are triangular in shape like the face of an octahedron, semi-rounded to elongated and a often the fragment piece of a diamond that has been cleaved.
Macles
These are odd shaped diamonds that may look like 2, three sided pyramids glued together at the base. The ribs are usually semi-curved or straight and the faces are usually grainy or wavy in texture. These shapes are somewhat of an oddity of nature as the cleavage planes in these roughs run parallel to the base rather than the octahedron faces. These are not commonly used for gem material as they are difficult to cut.
Surface Features
These are features found on the surface of a rough diamond regardless of the shape and are broken down into four categories as follows;
Mirror or Glass-like Surface
This type of surface can be found on any type of rough diamond and literally provides a ěwindowî that you can see into or through the diamond rough. These types of surfaces are common on the octahedron and dodecahedron shaped roughs. A feature that is often found on the surface of rough diamond, especially those with glass-like surfaces are Trigons. Trigons on diamonds are often quite visible to the naked eye. Trigons look like small triangles that are oriented 180 degrees from the octahedron faces that may be protruding or indented on the surface. They can appear on rough diamonds of any shape and are considered a strong indicator that the mineral is diamond. Image #7 is of a row of trigons at higher magnification. Having said this, the author has observed similar features on other minerals that form as octahedrons such as the mineral fluorite. It is likely that similar feature could be seen on any mineral of the cubic crystal system that forms in the shape of an octahedron. Image #8 is a highly magnified image of the surface of a fluorite in octahedron form that displays a trigon-like form on its surface.
Frosted or Waterworn Surface
As the name implies these types of surfaces are common to rough diamonds found in Placer deposits especially river bed deposits. These surfaces usually exist on non-octahedral and irregular shaped rough diamonds. This surface is often referred to as a sandblasted finish and displays varying amounts of transparency.
Grooved or Serrated Surface
These types of surface features are common to all types of rough diamonds shapes and include growth lines, wavy textured surfaces, and step like surfaces.
Growth lines are commonly observed at the edges of octahedron faces where the ribs are. As a diamond grows, new layers may grow over the old layers, however, sometimes the growth of the face ends just short of the rib and as such the edges begin to look layered at the ribs.(Image #9)
The same feature when observed on less well formed rough diamonds gives a more ěstep-likeî appearance. (Image #10) Rough Diamonds pg 40.
A rough diamond that has a ěwavyî appearance on its faces may have a more rounded faces and ribs.(Image #11) Rough Diamonds pg 38.
Blemished Surface
These features are caused by radiation from rock near the rough diamond that bombarded the diamond and ěstainedî the surface. The degree of staining may be as little as a spot or two, to completely covering the diamond. The staining is usually a dark green in color but may also have an appearance of brownish and grey tones.
Inclusions and Color
All diamonds possess some sort of inclusions. The visibility of inclusion within a diamond is simply based on magnification used to see them. Inclusions can be in many forms and usually visible to the naked eye and for those experienced in buying rough diamonds are often an aid to identifying a rough diamond and assessing its value. A few types of inclusions are; cracks that run through the diamond, hollow areas within the diamond or actual crystals of other material such as diopside, spinel or garnet to name a few. Generally speaking the more inclusions in the diamond rough the less valuable it is. However, even a heavily included diamond is valuable.
Color is more of a factor in the value of the diamond. Although rough diamonds may come in colors from clear to dark brownish to black, gem quality rough diamonds will appear colorless to varying shades of light yellow, some with very light tints of brown or greyish tones. These gem quality diamonds amount to about 10-30% of all diamonds mined*. The rest of the diamonds mined are used for industrial purposes such as sand paper grit or drill bits. Gem quality rough diamonds can also be pink, blue, red, green, or yellow. These ěfancyî colors are extremely rare. Rough diamonds mined in Canada are usually clear to very light yellow.
Methods of Identification
The first step to identifying a diamond rough is a visual examination. The rough itself may display several characteristic that are consistent with a rough diamond through its shape, exterior features and color. The use of a 10x power jewellers loupe is extremely handy at this stage as smaller surface feature may be quite apparent at 10x magnification. In addition to a visual examination, there are distinct characteristics of diamond that allow tests to be administered to accurately determine that it is a diamond. These tests as well as the visual characteristics that can be observed, together make for conclusive identification of diamond.
The characteristics of diamond that can be easily tested are its;
1) Hardness
2) Specific Gravity
3) Thermal Conductivity
Hardness
Diamond is the hardest known mineral, having a hardness of 10 on the Mohs hardness scale. The next hardest mineral is that of Corundum(Ruby or Sapphire) which has a hardness of 9. Other minerals that are considered quite hard are topaz and spinel which have a hardness of 8.
A simple test that has been used for centuries to test hardness is known as the scratch test. The test is done by scratching a mineral of unknown hardness against a mineral of known hardness, or visa versa. Quite simply, if the unknown mineral leaves a scratch mark, then it is harder than the other mineral. If the unknown mineral doesnít leave a mark then it is about the same hardness as the known mineral. And if the unknown mineral becomes scratched itself, then it is not as hard as the known mineral.
With this knowledge, and a small plate of corundum you can utilize this test. As corundum has a hardness of 9, if you are able to leave a small scratch in its surface with the unknown mineral then it can be said that the unknown mineral has a hardness greater than 9. In reverse, if you have an etching pencil with a small piece of corundum on the tip, it can be utilized in this way to try to scratch the surface of the unknown material.
Important:
1) It must be stressed that this test should never be applied to a cut and polished gem material. As the surface material of a diamond rough will be cut away during the cut and polish process, this concern is of less importance when applying this test to a rough diamond.
2) The second important point is that there may be a greater chance of damaging the unknown material when it is scratched against the known material. This is due to features such as cleavage planes or twinning planes. As such it is more advisable to utilize an etching pencil to try to make a small scratch in the surface of the unknown material. There is much less chance of damage to the unknown material when a etching pencil is used.
Specific Gravity
With the specific gravity of diamond being 3.52. This characteristic can be also be exploited through the use of heavy liquids. ěThe simplest procedure is to use a solution with the specific gravity of diamond = 3.52; then only a diamond will remain suspended, all other imitation stones will either sink or rise. Colorless topaz with a specific gravity of 3.53+/- 0.04 can be an exception. But in contrast to singly refractive diamond, topaz is doubly refractive.î Diamond Grading ABC pg 297. Single refraction or double refraction refers to a optical property of these gems that has not been examined in this paper. However, as was mentioned earlier, topaz has a hardness of 8, clearly separating it from diamond. Also, it does not fall into the cubic crystal system and as such would not be found in crystal shapes of octahedron or dodecahedron.
Thermal Conductivity
Diamonds are excellent conductors of heat. ěThe thermal resistance of a diamond is around one hundred times less than that of most other gemstones and imitations.îDiamond Grading ABC pg293. This characteristic can be exploited by a measure of how quickly a diamond disperses the heat of another object.
Simple, inexpensive instruments are available to test the thermal conductivity of diamond. They measure how quickly the heat is conducted by the change in temperature of the sensor. These instruments are quite accurate, compact and easy to use.
Note that these instruments were designed for the testing of cut and polished diamonds and in order for the instrument to work accurately the sensor tip should be placed on a clean flat surface of the diamond, perhaps a clear face on the gem, a ěwindowî, or cleavage plane. Placing the sensor tip on areas that display a grainy, gritty, or dirty surface may produce poor or inaccurate results. The author has used a Thermal Conductivity tester on rough diamonds with excellent results.
A simple and inexpensive kit can be assembled that can provide the tools to accurately identify a diamond. The kit should contain the following;
- 10x power jewellers loupe
- corundum plate(synthetic) and etching pencil
- vial of heavy liquid with a specific gravity of 3.52 at room temperature.
- thermal diamond tester
- jewellers tweezers or other tool to hold the specimen
Using information about the appearance of rough diamonds as well as tests that can be performed on them, an identification of rough diamond material can be made. The test that can be performed on the diamonds utilize the process of elimination. That is, if the material is harder than 9 on the Mohs hardness scale, it has a specific gravity of 3.52 then these two facts alone indicate the material would unlikely be any other material than diamond. Given a positive test on the thermal diamond tester only confirms the results. Lastly the visual examination may further confirm the fact findings through characteristic shapes or surface features found common to diamond roughs.
References
1) Rough Diamonds, A Practical Guide; Authored by Nizam Peters and Published by the American Institute of Diamond Cutting, U.S.A, 1998
2) Diamonds and The Northwest Territories, Canada; Published by the Department of Energy, Mines and Petroleum Resources, Mineral Division of the Government of the Northwest Territories, Canada, 1993 and reprinted in 1995
3) Diamond Grading ABC; Authored by Verena Pagel-Theisen G.G, F.G.A and Published by Rubin & Son bvba, Belgium, 1993
4) Gemmology; Authored by Peter G. Read and Published by Butterworth-Heinemann, U.S.A, 1991, second edition 1999.
5) Images #1, 2, 5, 6, 7, 8, and 9 are images of mineral samples from the authors collection and were taken buy the author through digital microscope.
|
Picture # 1 |
Picture # 2 |
Picture # 3 |
|
Picture # 4 |
Picture # 5 |
Picture # 6 |
|
Picture # 7 |
Picture # 8 |
Picture # 9 |
|
Picture # 10 |
Picture # 11 |